lumbar spine anatomy
*Check out my YouTube video on lumbar spine and disc anatomy for a less comprehensive, but quicker, review of this complex subject matter. Lumbar Spine and Disc Anatomy
The Nucleus | The Annulus | The Endplate | Lumbar Spine Nerves | Disc & Endplate Nerves
General Lumbar Spine & Disc Anatomy
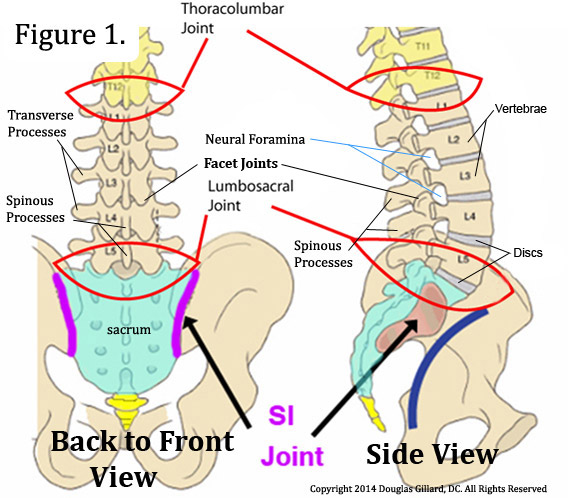 The lumbar spine (low back) is made up of five lumbar vertebrae (backbones) that are separated by five intervertebral discs (discs), the lowest of which sits on top of a large triangular shaped bone called the sacrum.
The lumbar spine (low back) is made up of five lumbar vertebrae (backbones) that are separated by five intervertebral discs (discs), the lowest of which sits on top of a large triangular shaped bone called the sacrum.
Posteriorly (toward the back), adjacent vertebrae (vertebrae next to one another) are connected together by two zygapophysial joints (facet joints or Z-joints) and a number of ligaments and muscles. (figure 1 & 2)
Through the center of each vertebra, as visualized in the axial view (akas: overhead view, transverse view or horizontal view), is a large bony hole called the vertebral foramen. Collectively, all of the vertebral foramen, in addition to the ligamentum flavum and posterior part of the discs, make up a structure called the vertebral canal (akas: spinal canal, vertebral canal). (figure 2)
The joining of two adjacent vertebrae, in the sagittal view (side view), forms the right and left neural foramina (akas: intervertebral foramina, IVF, neuroforamina), which is where spinal nerves exit the vertebral column (spine) on their way to innervate (give nerve supply two) the lower limbs (muscles, skin, bladder, blood vessels, you name it). (figure 2.5, & 10)
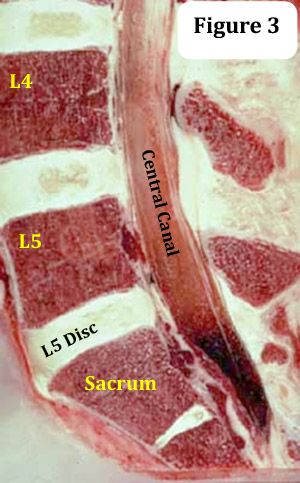
In the lumbar spine, the vertebral canal houses an extension of the spinal cord known as the thecal sac (aka, dural sac), which in turn houses the nerves (lumbar spinal nerves) that give life to the lower extremities, pelvis, and lower abdomen. It extends from the conus medullaris down to the second sacral segment (S2).
Technically, dura and arachnoid matter create the sac itself, and inside the sac is really the subarachnoid space which is filled with cerebrospinal fluid. The pia mater surrounds the individual traversing nerve roots of the cauda equina. In the center of the cauda equina is a slender filamentous structure made of glia and ependymal cells, the filum terminale, which extends from the conus medullaris (tip of the spinal cord), pierces the thecal sac at S2, and finally terminates at the coccyx where it blends in with the coccygeal connective tissue
Although not technically part of the spine, the semi-movable sacroiliac joints (SI joints) connect the two innominate (coxal or os coxa) bones (hip bones) to the sacrum and secure the pelvis posteriorly. It should be noted that the SI joint is a true diarthroidal joint which is well innervated (supplied by nerves). Although it doesn't happen often, it can become a source of chronic pain.
The smallest region of the spine is a triangular shaped bone called the coccyx (tailbone) which may be found coming off the most inferior (lowest) portion of the sacrum; it serves as an attachment point from muscles and ligaments.
A term that you should become familiar with is motion segment, which is the functional unit of the vertebral column. It consists of two adjacent vertebra, and a disc sandwich between them. The motion segment is held together by numerous muscles and ligaments, as well as the disc itself. For example, the second lowest motion segment of the spine, the L4/5 motion segment, is made of the L4 vertebra, the L4 disc, and the L5 vertebra.
*Although this page shall focus on lumbar spine anatomy, the thoracic and cervical spine share much of the same design and biomechanics; therefore, much of the discussion on this page may apply to those regions as well.
Axial Vertebra Anatomy
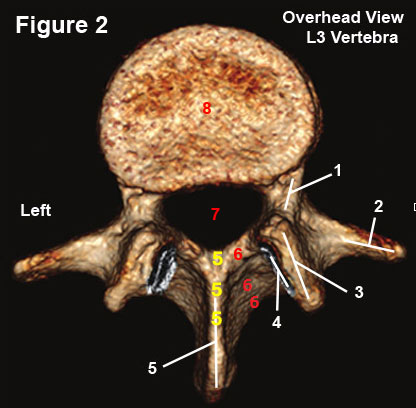 The lumbar vertebrae are very similarly designed and figure 2, which is a reconstructed CT axial image of the L3 vertebra, is quite representative of them all.
The lumbar vertebrae are very similarly designed and figure 2, which is a reconstructed CT axial image of the L3 vertebra, is quite representative of them all.
The vertebra has two basic parts: a front part that consists of the vertebral body (#8), and a slingshot-like back part, which is called the posterior arch.
The posterior arch, which is made up of two pedicles (#1), two laminae (#6) and a the base of a single spinous process (#5), is connected to the vertebral body by the pedicles—the strongest part of the vertebra. This connection creates the vertebral foramen (#7) which houses the thecal sac and its free-hanging lumbar nerve roots.
Arising from the junctions of the pedicles with the laminae are the transverse processes (#2) and the articular pillars which are made of the superior articular processes (#3) and the inferior articular processes (hidden below #3).
The articular processes connect adjacent vertebra at the zygapophysial joint (facet joint) and limit rotation and anterior translation (forward slip), which in turn protects the intervertebral disc from damage.
Note the groove for the inferior articular processes of L2 (#4) which, in combination with the superior a particular processes of L3, will create the L4 zygapophysial joint.
Sagittal Motion Segment Anatomy
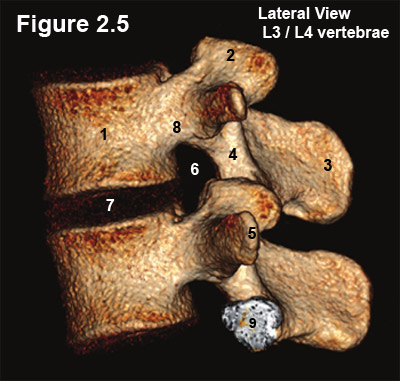 Figure 2.5 is a reconstructed CT image of a L3/4 motion segment, which demonstrates two vertebral bodies (#1) and a single disc (#7).
Figure 2.5 is a reconstructed CT image of a L3/4 motion segment, which demonstrates two vertebral bodies (#1) and a single disc (#7).
The joining of the two vertebrae creates a small bony hole on each side of the motion segment, which is called the neural foramen (#6) (foramina = plural). Specifically, its roof and floor are formed by the pedicles of the vertebra above and below (#8), its posterior (back) border is formed by the superior articular process (#2), and its anterior (front) border is formed by the posterolateral (back corner) of the vertebral body (#1) and disc (#7).
The Neural foramen is a very important structure, for it houses the exiting nerve root and the delicate dorsal root ganglion (DRG) which is where all of the sensory nerve cell bodies live. Sometimes, because of variation in anatomy, the DRG can be outside or even inside the neural foramen.
A constriction or narrowing of the neural foramen, which may occur via arthritic thickening (i.e., spondylosis, bone spur formation, or hypertrophic bone formation) of any of its bony walls or a special type of disc herniation (foraminal disc herniation), may result in a chronic-pain-producing condition called lateral stenosis. (Learn more)
The Intervertebral Disc
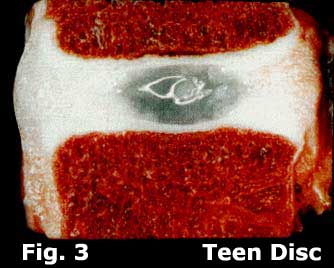
 The five discs of the lumbar spine are fibrocartilaginous structures that separate all of the lumbar vertebrae and are responsible for approximately two inches of overall height in the adult human. (figure 3)
The five discs of the lumbar spine are fibrocartilaginous structures that separate all of the lumbar vertebrae and are responsible for approximately two inches of overall height in the adult human. (figure 3)
Compared to the discs of the thoracic and cervical spine, the lumbar discs are taller and wider, yet more vulnerable to injury. This irony occurs because, in part, the discs are subject to a great range of motion while under great axial load (a downward force that is generated by gravity plus the weight of the body). Although the thoracic discs are also under significant axial load, the connecting rib cage dissipates much of that force and limits the range of motion.
Like the discs of the thoracic and cervical spine, the lumbar discs share one unique anatomical oddity: they are almost completely avascular (have no blood supply) and therefore rely upon primitive means (simple diffusion) for their nourishment and waste removal—this poor blood supply is the genesis for many of the causes of chronic low back pain.
There are three main anatomical regions in the disc: the gelatinous nucleus pulposus (nucleus), fibrous annulus fibrosus (annulus) and cartilaginous vertebral endplates (endplates).
Let's talk about each of them:
The Nucleus Pulposus
 The nucleus is a gelatinous (Jell-O like) material that normally resides in the center of the disc and is surrounded by the annulus and endplates. (figure 4)
The nucleus is a gelatinous (Jell-O like) material that normally resides in the center of the disc and is surrounded by the annulus and endplates. (figure 4)
One classic analogy describes the disc as the tire of a car: the annulus and endplates are the tough tread, and the nucleus is the air in its center, which is constantly trying to escape.
The cells of the nucleus produce mainly two types of molecules which create the substance of the nucleus: proteoglycan (65%) and type II collagen (17%).
The proteoglycans of the nucleus are particularly important for they, like tiny sponges, have a high affinity for water and are responsible for the fact that in the healthy young nucleus, the water content is an amazing 80%! [1,2] (figure 4) As we shall learn below, this high water content is needed for proper spine biomechanics.
Unfortunately, as the human ages, many of the nucleus cells (and annulus cells) die because of the poorly designed disc vascular system—they literally starve to death and/or are killed because of increased nucleus acidity. *With decreasing blood supply, the nucleus cells rely more heavily on anaerobic glycolysis for their energy production (like all cells, they need ATP to function), and the end product of anaerobic glycolysis is lactic acid.
As we shall learn on the degenerative disc disease (DDD) page, the decline of disc proteoglycans starts the train of DDD a rolling, which may lead to symptomatic annular tears, herniations, collapse and chronic disc-related pain.
The main functions of the nucleus include the transmission axial load to the vertebra below and to act as a intraspinal fulcrum (pivot-point) for all trunk motion. These functions are highly dependent upon the integrity of the annulus and endplates which must be able to resist or contain the nucleus's tendency to deform (squish or escape its confines) when compressed by axial load.
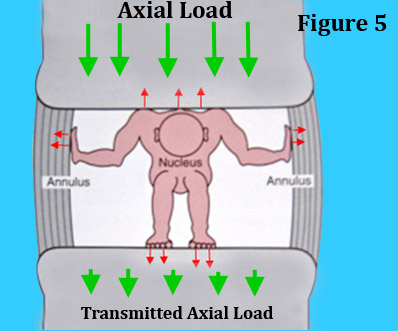 When functioning properly, the nucleus is a good example of what is called a closed hydraulic system, for the restrained water of the nucleus (by the annulus and endplates) becomes incompressible to the axial load and will in turn transfers these forces downward to the lower vertebra and set up the pivot-point. (figure 5)
When functioning properly, the nucleus is a good example of what is called a closed hydraulic system, for the restrained water of the nucleus (by the annulus and endplates) becomes incompressible to the axial load and will in turn transfers these forces downward to the lower vertebra and set up the pivot-point. (figure 5)
Figure 5 is a cartoon of what this closed hydraulic system looks like. Here we have a tiny man representing the nucleus who is trying to deform against the axial load (red arrows pushing outward in all directions); however, the immovable annulus and endplates are resisting. Therefore, since he cannot expand, he turns to stone which allows the axial load to be transmitted downward to the bottom vertebra.
As we will learn in the annular tear page, things go terribly wrong when the annulus fails to contain the nucleus, and the hydraulic system is destroyed.
The Annulus fibrosus
Although not as water-rich as the nucleus, the annulus still has a 65% water content because one of its cell types produces proteoglycans, just like the nucleus. In fact, the dry weight of the annulus has been calculated to be 20% proteoglycan. [1]
 The annulus has two types of cells: chondrocytes, which produce proteoglycans, and fibroblasts which produce predominantly type I collagen. Because of the type I collagen (which is the strongest of the 20+ types), the annulus is much stronger than the nucleus. [1,2]
The annulus has two types of cells: chondrocytes, which produce proteoglycans, and fibroblasts which produce predominantly type I collagen. Because of the type I collagen (which is the strongest of the 20+ types), the annulus is much stronger than the nucleus. [1,2]
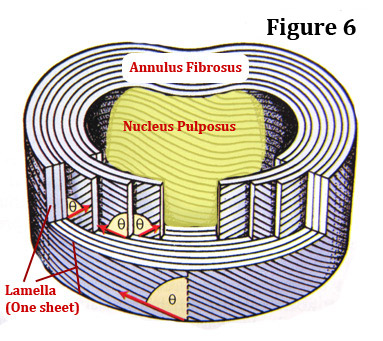
Like the plies in a tire tread, the annulus is made up of 10-20 belts of collagen called lamellae (lamella is singular), which all surround the nucleus in a layered fashion. (figure 6) Each lamella has a certain "grain" to it. In other words, the threads of collagen that makeup each lamella all run in the same direction.
For added strength, each adjacent lamella as its "grain" running in the opposite direction. For an example, the first lamella has its grain running in a positive slope, while the second lamella has its grain running in a negative slope. [3] (figure 6)
Unfortunately, many of the lamellae suffer gap defects and fail to completely surround the disc, which in turn creates a weakness—this is especially true of the posterior annulus, where strength is of the utmost importance. [3] These gaps defects may be one of the reasons that the posterior annulus is so vulnerable to the potentially painful annular tears and disc herniation.
As previously stated, the main function of the annulus is to contain the axial-load-related deformation of the nucleus, which in turn creates the closed hydraulic system and center pivot-point system for trunk movement.
The Vertebral Endplate
The endplates, which are positioned above and below the nucleus and most of the annulus, are thin (only 1mm) layers of hyaline cartilage that is made up of a familiar trio of molecule: proteoglycan, collagen and water. [4] Because of the biochemical similarity to the nucleus and the annulus, the endplates are in fact considered to be part of the disc and not part of the vertebral body. [2]
 Except for the very periphery of the disc, the nucleus and annulus are attached very firmly to the endplates and separation is difficult. However, the same cannot be said about the attachment between the endplates and the subchondral bone (aka, bony endplate) of the vertebral body. In fact, research has demonstrated that even fairly mild trauma to the lumbar spine can result in a separation of the endplate from the vertebra. [5]
Except for the very periphery of the disc, the nucleus and annulus are attached very firmly to the endplates and separation is difficult. However, the same cannot be said about the attachment between the endplates and the subchondral bone (aka, bony endplate) of the vertebral body. In fact, research has demonstrated that even fairly mild trauma to the lumbar spine can result in a separation of the endplate from the vertebra. [5]
The major function of the endplates is to act as a semi-permeable membrane [6] which allows nutrients and metabolites to diffuse into the disc from the capillary blood of the vertebral body and to allow waste products to diffuse out. It also prevents larger molecules, such as the all-important proteoglycan, from diffusing out of the annulus and nucleus.
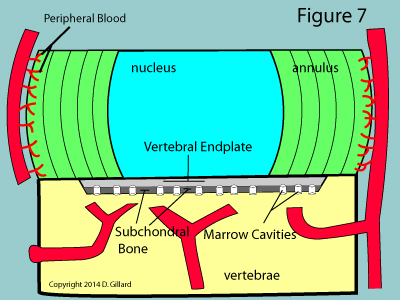
Such movement of molecules is absolutely mandatory for the survival of the disc, for as you may have noticed from figure 7, the disc tissue itself, except for the very periphery, is completely void of nutrient-giving blood vessels. Therefore, the cells of the nucleus and inner two-thirds of the annulus are completely dependent upon simple gradient-related diffusion of molecules in and out of the endplates. [2]
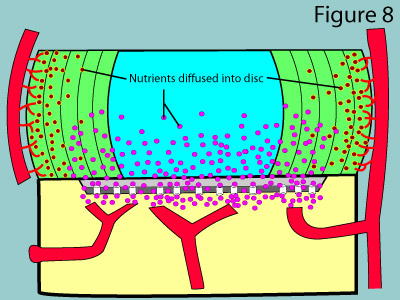 The astute reader may wonder how capillary blood can diffuse through the usually impermeable periosteum (the tough outer covering of the bone) as described above. Under normal circumstances, no diffusion would in fact occur; however, the subchondral bone of the vertebral bodies have special channels called marrow cavities which do in fact allow for diffusion to occur. (figure 7 & 8) Therefore, as long as these channels are open, the cells of the disc can get their needed oxygen, glucose, sulfur and and other nutrients, as well as have an avenue for waste removal.
The astute reader may wonder how capillary blood can diffuse through the usually impermeable periosteum (the tough outer covering of the bone) as described above. Under normal circumstances, no diffusion would in fact occur; however, the subchondral bone of the vertebral bodies have special channels called marrow cavities which do in fact allow for diffusion to occur. (figure 7 & 8) Therefore, as long as these channels are open, the cells of the disc can get their needed oxygen, glucose, sulfur and and other nutrients, as well as have an avenue for waste removal.
One of the proposed causes of degenerative disc disease (DDD) occurs when the marrow cavities become narrowed or closed from subchondral bone spondylosis (arthritis), which in turn starves the disc cells to death. (See the DDD page)
Although there have been many attempts at stabilizing or reversing DDD (via stem cell therapy, PRP injects, etc.), it is doubtful that these procedures will succeed in the long run because one of the main causes of DDD is the closing of the marrow cavities, a phenomena that is currently irreversible. [20]
Nerves of the Lumbar Spine
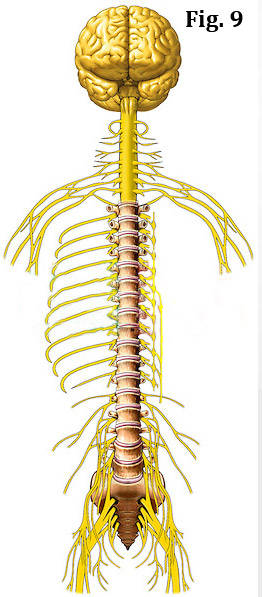 The spine is analogous to a superhighway that is enclosed by protective bony tunnels, all of which connect the brain to every nook and cranny of the body.
The spine is analogous to a superhighway that is enclosed by protective bony tunnels, all of which connect the brain to every nook and cranny of the body.
The brain, the master control center of the body, uses this superhighway to send out its commands as well as receive information about what's going on if the body. (figure 9) However, instead of using motor vehicles to send and receive information, the brain uses wire-like structures called nerves to get its job done. The nerves are actually made up of smaller nerves called to nerve fiber or axons.
The vast majority of these nerves, like the roots of a tree, leave the brain, course down the spine through a large central tunnel (central canal) and then exit the spine through smaller tunnels on the right and left (neural foramina).
The nerves that enter and leave the lumbar spine are extremely important, for they control the trunk and lower extremities with regard to motor function (the ability to move your muscles), sensory function (the ability to feel sensations such as touch, vibration, pain, and temperature) and autonomic function (the ability to control blood vessel size and sweating).
When an injury occurs to one or more of these nerves, the patient may suffer symptoms such as pain, loss of motor (muscle) function, loss of sensory (skin) function, or even loss of bladder/bowel function (a very serious condition called cauda equina syndrome). Sometimes the sympathetic component of the nerve root may become injured which may result in a very bizarre condition called chronic regional pain syndrome (CRPS) (formally called reflex sympathetic dystrophy or RSD) which may cause the lower extremity to swell, get very red and sensitive to the touch, and sweat.
In order to understand the different causes of chronic back and leg pain, we will need to study the three main nerve classifications: spinal nerves, lumbar nerve roots, and the sinuvertebral nerves.
Spinal Nerves
 The spinal nerve is formed by the convergence (coming together of) of the dorsal and ventral nerve root components of the exiting route and no longer is bathed in cerebrospinal fluid like the exiting nerve root is.
The spinal nerve is formed by the convergence (coming together of) of the dorsal and ventral nerve root components of the exiting route and no longer is bathed in cerebrospinal fluid like the exiting nerve root is.
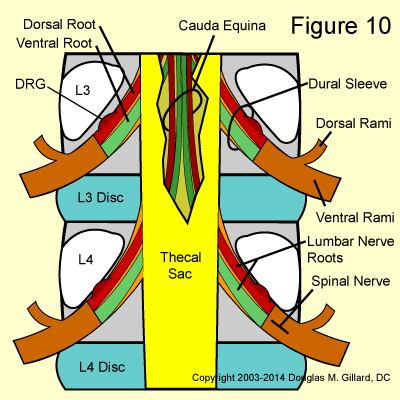
Also unlike the exiting root, the spinal nerve looses the protection of the dura mater as it morphs into perineurium which continues on to cover all peripheral nerves. (Figure 10)
Like the vast majority of peripheral nerves, the spinal nerve is called a mixed nerve, because it contains both sensory (afferent) and motor (efferent) nerve fibers. (It contains fibers of the autonomic system as well; however, this is beyond the scope of this page.)
Almost immediately after its emergence from the neural foramen, the spinal nerve diverges (splits) into two divisions: the dorsal ramus, which supplies sensation and motor function to the muscles and skin of the lower back and buttocks, as well as sensory function to the SI and facet joints; and the ventral ramus, which gives motor and sensory function to the muscles and skin of the lower extremities. (Figure 10) Nerves of the ventral rami form the lumbar and lumbosacral plexus which go on to form the important sciatic, femoral, and obturator nerves of the lower extremity. Furthermore, the ventral ramus gives off a small branch that helps form the sinuvertebral nerve (the nerve that transmits pain signals from of the back of the disc) and gives off another branch (gray ramus communicans) which connects it to the autonomic nervous system at the sympathetic trunk.
The dorsal ramus also has an important branch, the medial branch of the dorsal ramus, which gives the facet joints the ability to feel pain. This medial branch is the target of several different treatment interventions for chronic facet -related low back pain. (Figure14)
Lumbar Nerve Roots (aka: nerve roots)
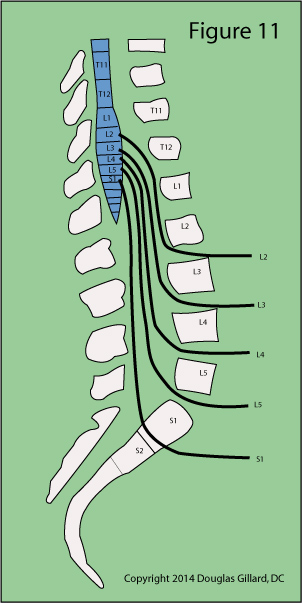 At approximately the T12 vertebral body level, the lumbar spinal nerves emerge bilaterally (from both sides) from the spinal cord and course downward within an extension of the spinal cord called the thecal sac (aka, dural sac) and then exit the spine at their respective neural foramina. (figure 10 & 11) The structure from which they arise, which is actually the end of the spinal cord, is called the conus medullaris and its most distal tip typically terminates at the L1 / L2 disc space. That's right, the spinal cord in the human fails to travel all the way down the spine! Instead, the nerve roots hang in a tough sac of dural matter called the dural sac or thecal sac.
At approximately the T12 vertebral body level, the lumbar spinal nerves emerge bilaterally (from both sides) from the spinal cord and course downward within an extension of the spinal cord called the thecal sac (aka, dural sac) and then exit the spine at their respective neural foramina. (figure 10 & 11) The structure from which they arise, which is actually the end of the spinal cord, is called the conus medullaris and its most distal tip typically terminates at the L1 / L2 disc space. That's right, the spinal cord in the human fails to travel all the way down the spine! Instead, the nerve roots hang in a tough sac of dural matter called the dural sac or thecal sac.
In reality, there are four lumbar nerve roots that are going to exit at each disc level: a right and left dorsal root (this carries sensory signals) and a right and left ventral root (this carries motor signals). (figure 10)
As the nerve roots approach their exiting neural foramina, they bud off the thecal sac at approximately the mid-vertebral body level of the same numbered vertebra (S1 is different and buds off at the L5 mid-vertebral body level).
For example, the L4 nerve root buds off the thecal sac at the mid L4 vertebral body level and, after rounding the pedicles, it leaves the spine through the L4 neuroforamina.
When the nerve roots bud off the thecal sac, they do not just break through it. Instead, they take with them the protective dura mater of the thecal sac which, along with the cerebrospinal fluid, stays with them all the way through the neural foramina. This protective extension of dura mater is called the dural sleeve. (figure 10)
Also remember that there are technically two lumbar nerve roots contained within each dural sleeve: a dorsal lumbar root and ventral lumbar root. However, clinicians and even spine surgeons virtually never make this anatomically-correct distinction and just call the them the exiting or traversing nerve root (see below).
The Exiting and Traversing Nerve Roots
Although the designations exiting and traversing nerve roots are typically not used by anatomists, clinicians, surgeons, and radiologists do use these terms frequently; therefore, it is important that you become familiar with them.
At each disc level, there are four vulnerable nerve roots: a right and left exiting root and a right and left traversing nerve root.
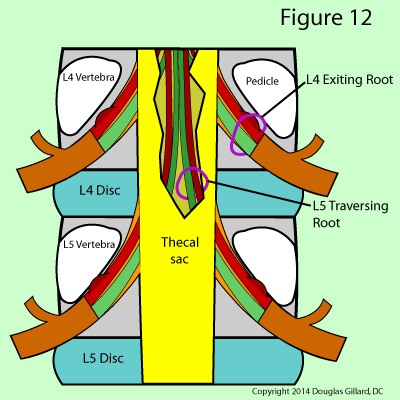 The exiting nerve root, as the name implies, is the one that has budded off the thecal sac and is on its way to and through the neural foramen. (figure 12) After its genesis, it will have the same number designation as the vertebral body, the disc it passes by, and the neural foramen it passes out of. For example, the left L4 exiting nerve root will bud off the thecal sac at the mid-vertebral body level of L4, wrap underneath the left L4 pedicle, pass by the left posterolateral aspect of the L4 disc, and proceed through the left L4 neural foramen before transforming into the left L4 spinal nerve. (figure 12)
The exiting nerve root, as the name implies, is the one that has budded off the thecal sac and is on its way to and through the neural foramen. (figure 12) After its genesis, it will have the same number designation as the vertebral body, the disc it passes by, and the neural foramen it passes out of. For example, the left L4 exiting nerve root will bud off the thecal sac at the mid-vertebral body level of L4, wrap underneath the left L4 pedicle, pass by the left posterolateral aspect of the L4 disc, and proceed through the left L4 neural foramen before transforming into the left L4 spinal nerve. (figure 12)
The traversing nerve root at the level of discussion is, in reality, the exiting nerve root for the segment below which has not quite budded off the thecal sac. In figure 12 you can see the traversing nerve root of L5 which is still contained within the thecal sac. For example, at the L4 disc level, we already know there is a L4 exiting nerve root that passes the posterolateral disc on its way to the neural foramen But there is also left L5 traversing nerve root positioned adjacent to the L4 disc (it is actually in a region of the central canal called the lateral recess); however, it is still within the thecal sac.
 The S1 nerve root is an exception to this rule as it typically buds off the thecal sac earlier (more cephalad [toward the head]) than the rest of lumbar nerve roots and is already in nerve-root form when it passes the L5 disc. I believe this makes the S1 nerve root particularly vulnerable to injury.
The S1 nerve root is an exception to this rule as it typically buds off the thecal sac earlier (more cephalad [toward the head]) than the rest of lumbar nerve roots and is already in nerve-root form when it passes the L5 disc. I believe this makes the S1 nerve root particularly vulnerable to injury.
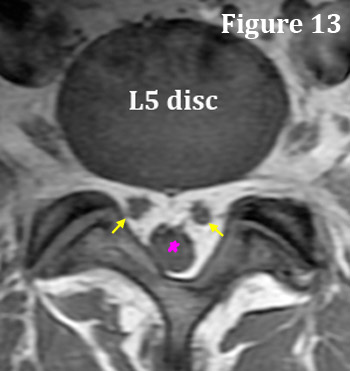
Don't believe me? This exception to the rule is easy to prove by looking at a typical T1-weighted axial MRI image of the L5 disc. In figure 13, you can clearly see that the S1 nerve roots (yellow arrows) are outside of the thecal sac (pink star); a phenomena that is not typically seen at any other disc level—rarely you will see it at L4.
I know this is a difficult topic to grasp; however, clinically speaking, it is extremely important information. If you can understand this, then I guarantee that you will know something 99% of all physicians don't (perhaps with the exception of spine surgeons).
Why is the stuff important? Because as we will learn on the disc herniation page, disc herniations can compress and irritate the traversing nerve root, the exiting nerve root, or both nerve roots! It all depends on the location and size of disc herniation.
Nerves of the Disc:
Did you know that the disc is wired for pain? [7] Surprisingly, despite the overwhelming scientific evidence, [8-11] very few physicians know or even care about this important fact.
In this section, we will discuss in great detail how pain signals are able to get out of the disc and ultimately up to the sensory cortex of the brain—the part of brain that translates these pain signals into the feeling of pain.
Learning the exact pathway of these pain signals is important, for if we know it, then we can target the pathway for treatment (glucocorticoid injections) or even destroy it (ablation) in order to mitigate chronic low back pain. Although the cause of these pain signals is beyond the scope of this page, I will say that tears through the annulus fibrosus (annular tears) are the usual suspect.
 Each region of the disc is innervated (supplied) by a different nerve (pain highway), which is a phenomena that oddly occurs in no other structure of the body. [19]
Each region of the disc is innervated (supplied) by a different nerve (pain highway), which is a phenomena that oddly occurs in no other structure of the body. [19]
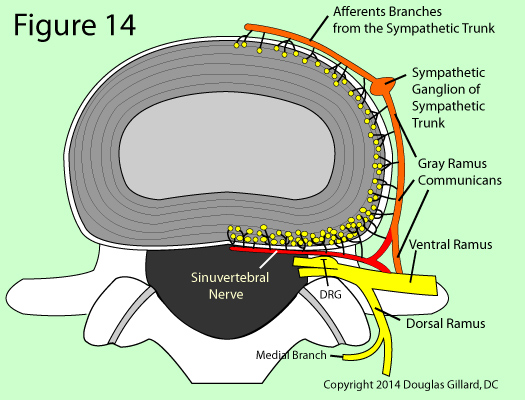
The posterior and posterolateral disc are innervated by the sinuvertebral nerve, the lateral disc by the gray ramus communicans (a sympathetic nerve of the autonomic system), and the anterior disc by sympathetic branches from the sympathetic trunk or ganglion. [2,10,15,16] (figure 14)
Another disc oddity is the fact that only the outer one-third (to a ~3 mm depth) of a healthy annulus is innervated and capable of transmitting pain. Therefore, the inner two-thirds of the annulus and the entire nucleus are completely avascular ( no blood vessels) and aneural (no nerves). [8,11,15,25]
The actual pain sensors of the disc (i.e., the free nerve endings of nociceptors), which are indicated by yellow dots in figure 14, are more heavily distributed in the posterior and posterolateral part of the annulus, as compared to the lateral or anterior regions. This probably means that the posterior and posterolateral disc are much more sensitive to annular tears (these regions can feel pain more easily) and the development of chronic back pain.
Since annular tears and, in some cases, their resultant disc herniations occur more frequently in the posterior and posterolateral regions of the disc, we shall focus our study on the principle nerve that carries pain signals away from this area, the sinuvertebral nerve.
The Sinuvertebral Nerve: "Oh what a tangled web we weave...." Sir Walter Scott (Marmion, 1808)
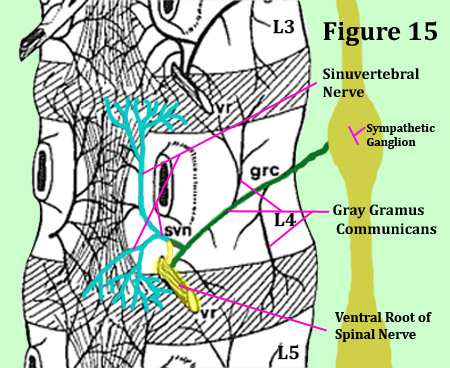
The bilateral sinuvertebral nerve may be considered a conjoined nerve (like a wishbone), for it arises from both the ventral ramus of the spinal nerve (a somatic nerve) and the gray ramus communicans of the sympathetic ganglion (an autonomic nerve). [16,25] (figure 14 & 15) Not only does this important nerve innervate the posterior outer one-third of the annulus at the same level, it also innervates the same distribution (parts of the disc) at the level above! (figure 15) [2,10,16,17] For an example, the L4 sinuvertebral nerves innervates the posterior annulus of L3 and L4.
To make things more confusing, there is other research showing that a single sinuvertebral nerves innervates three disc levels! [25,29]
So what does that mean? It means at the very least, there is no way the human can normally tell the difference between, for example, an L3 versus an L4 painful disc, for they are both wired together. (*Only with a special test called provocative discography could such a distinction be made.)
 We know that the sinuvertebral nerve carries pain signals out of the posterior region of the disc and that these signals eventually make it up to one (or possibly more) of the lumbar segments of the spinal cord (these segments are called myelomeres). Thereafter, the signals cross-over (decussate) to the opposite side of the cord, are transmitted up to the thalamus of the brain, and then reach the sensory cortex.
We know that the sinuvertebral nerve carries pain signals out of the posterior region of the disc and that these signals eventually make it up to one (or possibly more) of the lumbar segments of the spinal cord (these segments are called myelomeres). Thereafter, the signals cross-over (decussate) to the opposite side of the cord, are transmitted up to the thalamus of the brain, and then reach the sensory cortex.
However, notwithstanding a tremendous amount of research, [10,11,16,17,18,22-24] we still don't know the exact pathway that these pain signals take after they leave the sinuvertebral nerve. This is unfortunate and greatly hampers any attempt at treating or eliminating the nerves that carry such pain signals.
Logically, you would think that the pain signals from, for example, the L5 disc would simply take the shortest path to the DRG, which in this case would be from the sinuvertebral nerve to the same-level ventral ramus and then to the same level DRG. (*Note: as mentioned above, once pain signals reach the DRG, we know the exact path they take to the sensory cortex of the brain). However, there is fairly strong scientific evidence (albeit much of it is from animal study) which demonstrates this simple route is not employed (used). [21-24]
Instead, it seems that pain signals from all of the lumbar discs (we will use L4 for this example) roughly take the same major detour after they leave the sinuvertebral nerve:
The sinuvertebral nerve to the gray ramus communicans (GRC) --> the GRC to the sympathetic ganglion of L4--> the sympathetic ganglion of L4, up to the sympathetic ganglion of L1 or L2 --> the sympathetic ganglion of L1 or L2, across the white ramus communicans (WRC) (which are only present at L1 and L2) --> the WRC to the same level ventral root of the spinal nerve of L1 or L2--> the ventral root of L1 or L2 to the DRG of L1 or L2 --> DRG of L1 or L2 to the spinal cord at the L1 or L2 myelomere --> spinal cord to the sensory cortex. [22-24]
While the foregoing pathway could be semi-logical for the L1 or L2 disc, it sure doesn't make a whole lot of sense for the L4 or L5 disc, because such a route is so far off the short and simple path that it is almost silly, yet this seems to be reality.
Other high quality research (although it too was animal research) paints an even more convoluted picture, for it suggests that pain signals from one disc actually end up in multiple DRGs, none of which are the DRG of the disc that was actually generating the pain signals in the first place. [18]
There is actually one human study, however, (completed by a well-respected Japanese Medical school) that seems to confirm the animal studies and is worthy of a quick discussion. *However, please note that this investigation was only mediocre in design; therefore, the results should taken with a grain of salt.
Nakamura (1996): Strange Routing of L4 or L5 Disc Pain
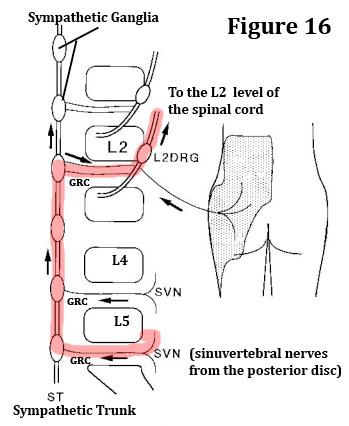 In 1996, Nakamura et al. [19] published the results of their one-of-a-kind investigation which tested the hypothesis that discogenic low back pain from the lower lumbar spine (L4 and L5) conveyed pain signals to the spinal cord and brain through the upper lumbar spine (L2 dorsal root ganglion), which effectively bypassed the actual source of the pain ( i.e., the L4 or L5 disc).
In 1996, Nakamura et al. [19] published the results of their one-of-a-kind investigation which tested the hypothesis that discogenic low back pain from the lower lumbar spine (L4 and L5) conveyed pain signals to the spinal cord and brain through the upper lumbar spine (L2 dorsal root ganglion), which effectively bypassed the actual source of the pain ( i.e., the L4 or L5 disc).
METHODS: A group of 33 chronic low back pain patients, all in whom shared the MRI-confirmed diagnosis of a symptomatic L4 or L5 annular tear or herniation, were gathered and treated with a selective nerve root block—a fluoroscopically guided small-volume (1.5 mL) injection of lidocaine (a powerful anesthetic), which was released at the entrance of the neural foramen (i.e., in very close to the DRG).
*The theory here is simple: if pain signals from the L4 or L5 disc are being transmitted through the L2 DRG, then the application of lidocaine onto the L2 DRG should temporarily block those pain signals and the patient will feel a significant lessening of the pain, as well as an increased ability to function.
RESULTS: Within 15 minutes following the selective nerve root block, all of the patients experienced at least some reduction in low back pain (P<0.001) and had an increased ability to remain flexed at the waist in a standing position (this is called the static flexion test and is quite difficult for a disc patient to perform). Furthermore, 79% of the group experienced a complete or near complete reduction of low back pain.
CONCLUSION: The authors concluded by saying, "...discogenic low-back pain is transmitted non-segmentally by visceral sympathetic afferents mainly through the L2 spinal nerve root...." and "Discogenic low-back pain may be a type of visceral pain." [19] Visceral pain, such as a tummy ache, can be quite vague and diffuse and has a tendency to shift around a bit in location—some familiar?
How Do We Know for Sure?
How do we know low back pain signals are not transmitted via the simple route described previously: same-level disc--> sinuvertebral nerve--> same-level ventral ramus--> same-level DRG?
Although in this day and age we could probably never get away with it (institutional review boards are now mandatory and very strict about the type of human experimentation that is allowed), there have been some human studies performed during disc surgery while the patient was awake (under local anesthetic)! For an example, in one famous study which was done during the discectomy procedure it was demonstrated that by anesthetizing the inflamed exiting nerve root at the same level of the disc herniation (i.e., the one causing the sciatica), the patient would feel relief of the leg pain but no relief of the low back pain (back pain often occur together). Therefore, the logical conclusion was/is that the back pain signals were/are not traveling through the anesthetized, same-level nerve root, for if they were, the back pain would have went away.
Furthermore, we also know from the results of modern transforaminal epidural steroid injections (TFESIs) and selective nerve root blocks (SNRBs), that anesthetizing the spinal nerve (which is adjacent/distal to the DRG) will also have very little effect, if any, on a patient's low back pain—these types of procedures are for the treatment of radicular pain (sciatica, lower extremity pain) only.
Therefore, based upon the scientific and anecdotal evidence, we are fairly confident that pain signals from an injured disc do not reenter the DRG at the same level and are actually handled by sympathetic-nerve-rerouting to DRGs at higher levels.
In closing, you may be asking, "what does all this mean?" It means that more research needs to be done and more funding needs to be made available for elucidating the pathways of discogenic pain, for until we know these pathways for sure, patients will continue to suffer chronic low back pain without much hope of a noninvasive cure.
The Basivertebral Nerve: Vertebrogenic back pain?
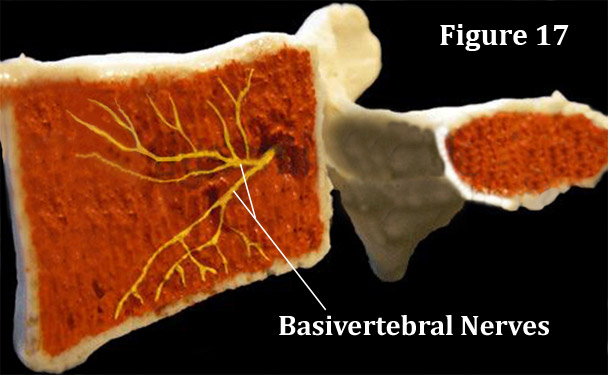 Over the past decade a new diagnosis has arisen with regard to chronic intractable low back pain: Vertebrogenic pain syndrome (VPS).
Over the past decade a new diagnosis has arisen with regard to chronic intractable low back pain: Vertebrogenic pain syndrome (VPS).
VPS occurs when the vertebral endplates of the disc and the associated subchondral bone of the vertebrae (which are not very thick structures and are actually movable when pressurized) become irritated, inflamed and start to generate chronic pain signals.
The science behind this new pain syndrome is not yet very strong, but does exist.
Basically there are two published investigations, both of which demonstrating that the basivertebral nerves of the vertebral body gives off branches that supply the subchondral bone of the vertebral endplates [6] with pain-carrying fiber. [6,28] Furthermore, we know from the work of Bogduk [15,16] that the basivertebral nerves leave the basivertebral foramen and turns into divisions of... you guessed it... our friend the sinuvertebral nerves. From there we believe that the pain signals through the gray ramus communicans, to the sympathetic trunk, probably up the sympathetic trunk, down the grey or white ramus communicans to the ventral root of the spinal nerve, into the dorsal root ganglia (at God only knows what level) and up to the brain via familiar pathways.
Treatment for chronic VPS is limited to attempts at either injecting a steroid into the basivertebral foramen or ablating the nerves exiting that foramen. I still can't find any papers reporting the outcomes of either procedure, but I'm sure they will be coming soon.
References
1) Beard HK, et al. Biochemical changes in the intervertebral disc. In: Jayson MIV, ed. The Spine and Backache, 2nd edn. London: Pitman;1980:407-436.
2) Bogduk N, et al. The interbody joint and the intervertebral disc. In: Bogduk 4th, ed. Clinical anatomy of the lumbar spine and sacrum. London: Elsevier; 2008:11-28.
3) Marchand F, et al. Investigation of the laminate structure of the lumbar disc annulus fibrosus. Spine 1990;15:402-410.
4) Roberts S, et al. Biochemical and structural properties of the cartilage endplate and its relation to the intervertebral disc. Spine 1989;14:166-174.
5) McNabb I. Backache. Baltimore: Williams and Wilkins;1977:4-7.
6) Bailey JF, et al. "Intervention patterns of PGP 9.5-positive nerve fibers within the human lumbar vertebra. J. Anat. 2011;218:263-270.
7) Bogduk N, Aprill C, Derby R. lumbar discogenic pain: state-of-the-art review. Pain Med. 2013;14: 813-836.
8) Malinsky J. The ontogenetic development of nerve terminations in the intervertebral disc of man. Acta Anat 1959;38:96-113.
9) Yoshizawa H, et al. The neuropathology of intervertebral discs removed for low back pain. J Path 1980;132:95-104.
10) Bogduk N. The lumbar disc and low back pain. Neurosurg Clin N Am 1991;2:791-806.
11) Suseki K. Sensory fibers from lumbar intervertebral discs pass-through rami communicantes. J Bone Surg Br. 1998;80-B:737-742.
12) Higuchi K, Sato T. Anatomical study of lumbar spine innervation. Folia Morphol (Warsz) 2002;61:71-79.
13) Takebayashi T, et al. Sympathetic afferent units from lumbar intervertebral discs. J Bone Joint Surg Br 2006;88-B;554-7.
14) Schwarzer AC, Aprill CN, Derby R, Bogduk N, Kine G. The prevalence and clinical features of internal disc disruption in patients with chronic low back pain. Spine 1995;20:1878-1883.
15) Jackson HC, et al. Nerve endings in the human lumbar spinal column and related structures. J Bone Joint Surg AM 1966;48-A:1272-82.
16) Bogduk N, et al. The nerve supply to the human lumbar intervertebral discs. J Anat 1981;132:39-56.
17) Bogduk N. The innervation of the lumbar spine. Spine 1983;8:286-93.
18) Ohtori S, et al. Sensory innervation of the dorsal portion of the lumbar intervertebral discs in rats. Spine 2001;26:946-950.
19) Nakamura SI, et al. Origin of nerves supplying the posterior portion of the lumbar intervertebral discs in rats. spine 1996;21:917-924.
20) Grunhagenn T, Urban JPG, et al. Nutrient supply and intervertebral disc metabolism. J Bone Joint Surg AM 2006;88A:30-35.
21) Nakamura SI, et al. The afferent pathways of discogenic low back pain. J Bone Joint Surg Br 1996;78B:606-612.
22) Cavanaugh JM, et al. Mechanisms of low back pain: a neurophysiological and neuroanatomical study. Clin Orthop 1997;335:166-180.
23) Goren GJ et al. The human sinuvertebral nerves. Annals R Coll Surg 1998;70:167.
24) Goren GJ et al. Nerves and nerve plexuses of the lumbar vertebral column. Amer J Anat 1990;188:282-296.
25) Edgar MA, Nundy S. Innervation of the spinal dura mater. J Neurol Neurosurg Psychiat 1966;29:530-534.
26) Kuslich SD, et al. The tissue origin of low back pain and sciatica. Clin North Am 1991;22:181-187.
27) Walsh TR, et al. Lumbar discography in normal subjects. J Bone Joint Surg 1991;72A:1081-1088.
28) Fras C, et al. Substance P- containing nerves within the human vertebral body: an immunohistochemical study of the basivertebral nerve. Spine J 2003;3:63-67.
29) Stillwell DI Jr. The nerve supply of the vertebral column and its associated structures in the monkey. Anat Rec 1956;125:139-169.
